
Halocarbons are industrially produced gases whose main use is as refrigerants in fridges, freezers and in air conditioning systems. The largest source of halocarbon emissions is commercial refrigeration, including food storage [1]. Although refrigerant gases are emitted in relatively small quantities compared to CO2 and methane, due to their chemical structure they have a much higher global warming potential, hence their relatively large contribution to global warming, accounting for 11% of all anthropogenic radiative forcing, according to the latest IPCC data [2].
Currently official national greenhouse gas inventories exclude all CFCs and HCFCs even though they are responsible for approximately 85% of the global warming from halocarbons [2] . This is because they were assumed to be dealt with by agreements to phase out ozone depleting chemicals. However, this has failed to tackle the problem and overall, they continue to be a menace both to the climate and the ozone layer.
CFCs still at high atmospheric levels.
Although use of CFCs in fridges has largely stopped because of their destructive impact on the Ozone layer, they remain at a high level in the atmosphere due to their long atmospheric lifetime (see Figure 1 and 2). CFC-12 has a Global Warming Potential (GWP) of 11,000 and atmospheric lifetime 100 years) . This potent CFC is still having by far the highest global warming impact of any of the halocarbon refrigerant gases[2] and still has an atmospheric concentration of over 500 parts per trillion (ppt) in 2010 [3]. As shown in Figure 1, atmospheric concentration of CFC-12 was higher in 2010 than in 1989, the year the Montreal Protocol was first enforced. CFC-113 was also at a relatively high level in 2010, at over 75 ppt, as shown in Figure 2.
Figure 1: CFC-12 Atmospheric levels from 1977-2010 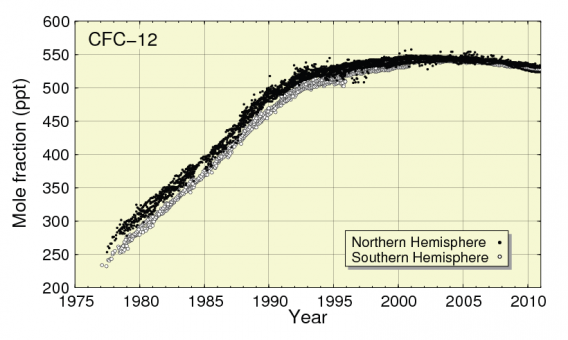
Source JMA & WMO, 2012 [3]
Figure 2: CFC-113 levels from 1982 -2010 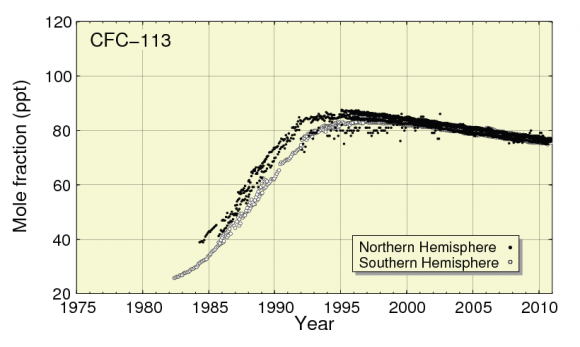 Source JMA & WMO, 2012 [3]
Source JMA & WMO, 2012 [3]
Solving the CFC-113a ‘mystery’
Recent research has shown atmospheric levels of 3 other types of CFC to be increasing, with CFC-113a being the highest of these. levels of CFC-113a (otherwise known as R113a) had reached 0.48 ppt in 2012 [4] more than doubling in the two years since 2010 and it is still rising at an accelerating rate [5] . Several recent news reports have discussed this, saying the source of CFC-113a is a mystery [5, 6]. However, loopholes in the Montreal Protocol allow it to be used as an intermediate chemical in the manufacture of other commonly used chemicals, including (HFC-134a) the most popular CFC refrigerant gas [7] . It is also used as a precursor in the manufacture of flouro-pesticides [8].
Markets for CFC-113a
There were several online adverts for CFC-113a supplies, such as on the website, Alibaba.com [9,10]. However, some of these seem to have been taken down since I found them on March 10 2014. However, the chemical is still for sale, listed as R113a. The large minimum volume for orders from a major manufacturer of 1140 units of 200L canisters in one advert (which was still up on March 11, 2014) [10] would inevitably increase the chances of excess unused product escaping to the atmosphere if not properly disposed of. This minimum order size is 228,000 litres of CFC-113a. Another supplier of CFC-113a based in China [9] has 46% of its consignments going to markets in China, with the Middle East being the second biggest market (see Figure 3).
Figure 3: CFC-113a customers (by region) from a major global supplier 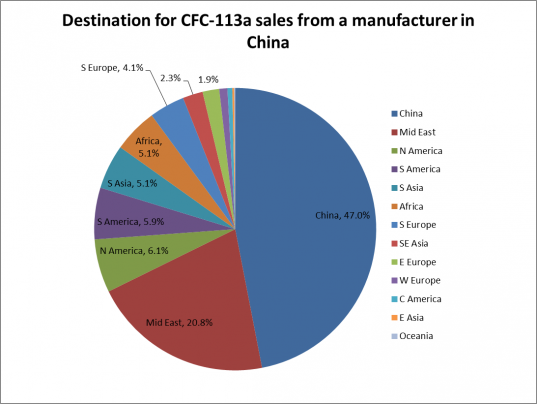
Source: Alibaba.com [9]
The atmospheric level of CFC-113a is approximately 1000 times lower than CFC-12 levels, and 140 times lower than CFC-113, so is still relatively insignificant in it’s impact compared to these other ozone depleting greenhouse gases.However, the rise of use and atmospheric levels of R-134a, of which CFC-113a can be used for production, will lead to a further rise in CFC-113a levels.
Use of CFCs for pesticide production
The use of CFC-113a in the manufacture of pesticides [8] is also of concern, as global warming is likely to increase the threat to agricultural crops from pathogens and pests, hence increasing the use of synthetic pesticides to attempt to deal with this increasing problem. Pesticide manufacture will therefore be another growing source of CFC pollution unless loopholes are closed in international and national legislation.
HCFCs on the rise
HCFCs are a type of halocarbon gas that damages the ozone layer as well as causing significant climate impacts, and is still legal to be used as a refrigerant, but is supposed to be phased out by 2030. In 2006, more than half of all commercial refrigerants were HCFCs [11]. See figure 4 below:
Figure 4: Distribution of Global Commercial Refrigerant Stock by Refrigerant Type (2006) 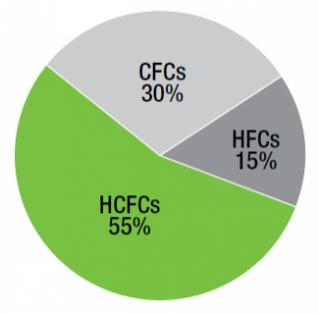
Source: EPA, 2010 [22]
Despite policy efforts to gradually phase them out due to their impacts on the ozone layer, continued production of HCFCs has led to their atmospheric level doubling in the last decade and their production is still expected to continue increasing to 2030, particularly in developing countries such as China where there is rapidly increasing demand for refrigeration [11]. HCFC-22, the most popular HCFC refrigerant, has a 20 year GWP of 5160 and its production leads to the emission of HFC-23 which is an even more potent greenhouse gas, with a 20 year global warming potential of 12,000. As shown in Figure 5, HCFC-22 had reached a level of over 220 ppt in 2010 (an atmospheric concentration over 450 times greater than CFC-113a) and continues to rise rapidly.
Figure 5; Rapidly increasing atmospheric levels of HCFC-22 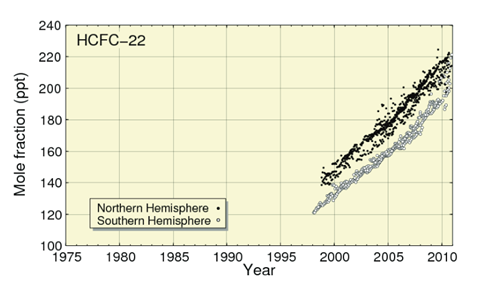
Source: JMA & WMO,2012 ![]()
As shown in Figure 6, atmospheric levels of HCFC-142b have almost tripled between 1995 and 2010, increasing from around 7ppt to over 21ppt
Figure 6: Increase in atmospheric levels of HCFC-142b 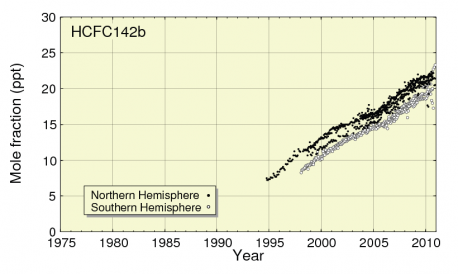
Source: JMA & WMO,2012
The newly discovered HCFC-133a, featured in the recent paper [4] and associated news stories, reached 0.37ppt, an atmospheric concentration approximately 600 times lower than levels of HCFC-22 and 60 times lower than levels of HCFC-142b.
The Rise in HFCs – Refrigerant leakage from supermarkets
There were around 530,000 supermarkets worldwide in 2006, using approximately 550,000 tonnes of refrigerant [11].The average refrigerant leak rate is 10% per year [12]. The estimated total amount of emissions from HFCs (not including HCFCs) used in commercial refrigeration in 2010 was 346Mt CO2e [11]. This is likely to have increased substantially since, and is likely to continue to do so, particularly in non OECD countries with increasing demand for refrigeration [1]. The most common supermarket HFC refrigerant, R404a, has a 20 year Global warming Potential (GWP) of 6010, meaning it is over 6000 times more effective at trapping heat in the atmosphere than CO2 over a twenty year period. Therefore, the emission of just 1 kilogram of this refrigerant causes the equivalent global warming effect of driving a 2 litre Skoda Octavia for 25,000 miles [12].
Rapid rise in HFC-134a use.
Use of HFC-134a, a popular refrigerant and a constituent of R404a, has also been rapidly increasing as CFCs are replaced. Atmospheric levels of this gas have increased by over 400% in the last decade [3].This refrigerant gas has a 20 year GWP of 3710. Global production of HFCs is set to increase five-fold by 2030, mainly due to increasing demand for refrigeration in developing countries [1].The rise in its production could also account for the rise in CFC-113a, which can be used as a precursor chemical in its manufacture. The rapid rise in atmospheric levels of HFC-134a is shown in Figure 7.
Figure 7: Rapid rise in atmospheric HFC-134a between 1995 & 2010 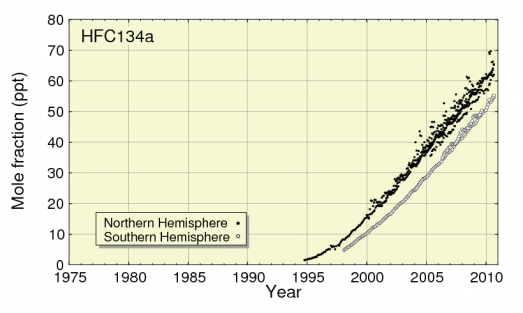
(JMA, WMO, 2012)
Conclusion
Any potential climate benefits from the phasing out of CFCs are being cancelled out by increasing demand for replacement gases which are also adding to global warming, both directly, through the rise in HCFCs and HFCs as replacements, and indirectly by the use of CFCs, in the manufacture of other refrigerants as well as pesticides. Blog by Daniel Kieve.
[1] EPA Global Anthropogenic Non-CO2 Greenhouse Gas Emissions: 1990 – 2030, US Environmental Protection Agency, December 2012
[2] IPCC Working Group I Contribution To The IPCC Fifth Assessment Report CLIMATE CHANGE 2013: THE PHYSICAL SCIENCE BASIS , IPCC, 2013 http://www.climatechange2013.org/images/uploads/WGIAR5_WGI-12Doc2b_FinalDraft_All.pdf [3] Halocarbons And Other Halogenated Species, Japan Meteorological Agency & WMO, 2012 http://ds.data.jma.go.jp/gmd/wdcgg/pub/products/summary/sum36/24_06haloc.pdf
[4] Newly detected ozone-depleting substances in the atmosphere Johannes C. Laube, Mike J. Newland, Christopher Hogan, Carl A. M. Brenninkmeijer et al, 2014, Nature Geoscience (2014) doi:10.1038/ngeo2109
[5] Four banned ozone depleters detected in the atmosphere, New Scientist article, 09.03.2014 http://www.newscientist.com/article/dn25188-four-banned-ozone-depleters-detected-in-the-atmosphere.html
[6] New ozone-destroying chemicals found in atmosphere Mysterious compounds undermining recovery of giant ozone hole over Antarctica, scientists warn http://www.theguardian.com/environment/2014/mar/09/ozone-hole-antarctica-chemicals [7] SELECTION OF COMMERCIAL PROCESSES FOR HFC-134a AND HFC-125 USING CORE CONCEPTS OF CATALYSIS, EQUILIBRIUM, LAW OF MASS ACTION, KINETICS AND THERMODYNAMICS http://chemcases.com/fluoro/fluoro14.htm
[8] Trifluorotrichloroethane CFC113a pesticides intermediate Advertisement: http://135510.zj.cn.hg.customer.hi2000.com/pages/pro-e.htm
[9] Hangzhou Sibolan Heating And Refrigerating Equipment Co., Ltd Advert on Alibaba.com website http://www.alibaba.com/product-detail/cfc-113a_916003123.html
[10] Tichlorotrifluoroethane R113a, Zhejiang Kingding Machinery Technology Co., Ltd advert on Alibaba website http://www.alibaba.com/product-detail/Tichlorotrifluoroethane-R113a_703144336.html
[11] Transitioning To Low-GWP Alternatives In Commercial Refrigeration (2010), U.S. Environmental Protection Agency (EPA), USA http://www.epa.gov/ozone/downloads/EPA_HFC_ComRef.pdf
[12] Development and demonstration of a prototype transcritical CO2 refrigeration system, LIFE05 ENV/DK/000158, EC,2007,http://ec.europa.eu/environment/life/project/Projects/index.cfm?fuseaction=home.showFile&rep=file&fil=LIFE05_ENV_DK_000156_LAYMAN.pdf
